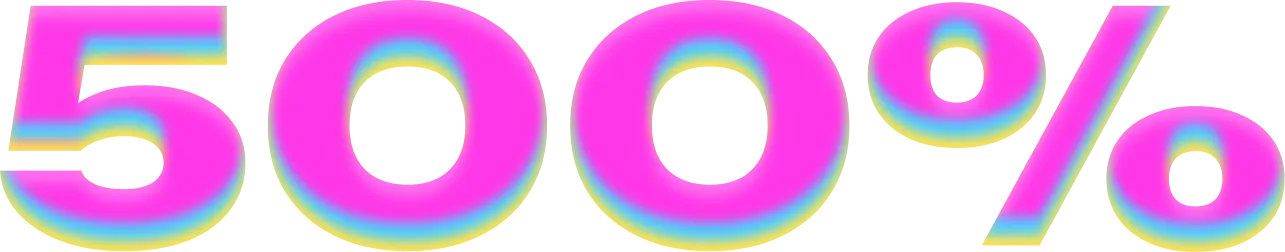Казино 1вин (1Win)
Казино 1вин (1Win) — это современная онлайн-платформа, которая сочетает в себе надежность, лицензию, обширный выбор игр и быструю техническую поддержку. С момента запуска 1Win зарекомендовало себя как одно из самых популярных казино для русскоязычных игроков. Здесь представлены тысячи автоматов, live-игры с реальными дилерами, краш-раздел и ставки на спорт. Простая регистрация, рабочее зеркало для обхода блокировок и моментальные выплаты делают 1вин идеальной точкой входа в мир азартных развлечений онлайн.
| 🥇 Название | 1вин |
| ⬆ Год запуска | 2018 |
| ✅ Лицензия | Curacao OGL/2023/964/0408 |
| 🎰 Основные разделы | Слоты, live casino, crash-игры, ставки |
| 🎁 Бонусы | до 500% + 250 фриспинов |
| 🎟 Промокод | 1WINCASINO |
| 📲 Устройства | Android, iOS, ПК, планшеты |
| 💳 Оплата | Visa, Mastercard, СБП, QIWI, Piastrix, криптовалюта |
| 💱 Валюты | RUB, USD, EUR, BTC, USDT |
| ⬇️ Минимальный депозит | 100 RUB |
| ⬇️ Минимальный вывод | 500 RUB |
| 📞 Поддержка | 24/7: чат, Telegram, email |
Что предлагает Казино 1вин (1Win)?
1Win предлагает обширный выбор слотов, игр с живыми дилерами, раздел crash-игр и ставки. Всё доступно на русском языке и с мгновенными выплатами.
Какие бонусы можно получить в 1вин Казино?
Новые игроки получают до 500% к первым пополнениям и до 250 фриспинов. Промокод 1WINCASINO активирует приветственный бонус.
Есть ли зеркало у 1вин Казино?
Да, для обхода блокировок используется зеркало 1Win. Оно предоставляет полный функционал основного сайта без использования VPN.
Можно ли играть на 1Win с телефона?
Да, 1вин Казино полностью адаптировано под мобильные устройства и предлагает приложения для Android и iOS с полным функционалом.
Как быстро происходят выплаты на 1Win?
Вывод выигрышей занимает от 15 минут до нескольких часов. Криптовалютные и Piastrix-транзакции обрабатываются особенно быстро.
Poker
Получите бонус
Участвуйте, играйте и выигрывайте